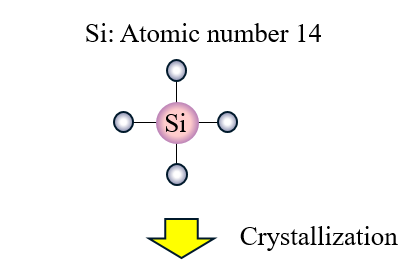
Semiconductor material…Silicon
A silicon atom has 14 electrons around the nucleus, and of these, there are 4 valence electrons on the outermost orbital. When this is made into a single crystal, it can be used as a material for semiconductor products.
-
-
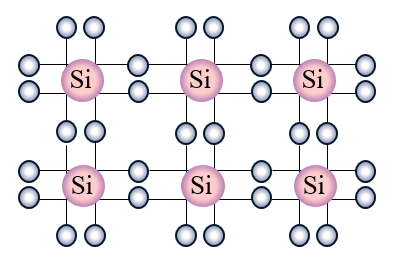
- Forming crystals by bonding silicon atoms in a regular structure
-
When it crystalizes, the nuclei share electrons and they bond with 8 electrons around each nucleus. Electricity for the most part does not conduct in this pure monocrystalline silicon state.
Doping silicon with other impurities changes it so it is conductive. The semiconductor is categorized as a p-type or n-type depending on the type of impurities that are doped. Junctions based on the p-types and n-types are integrated into one chip in order to use it as an electronic component.
p-type semiconductor and n-type semiconductor
-
-
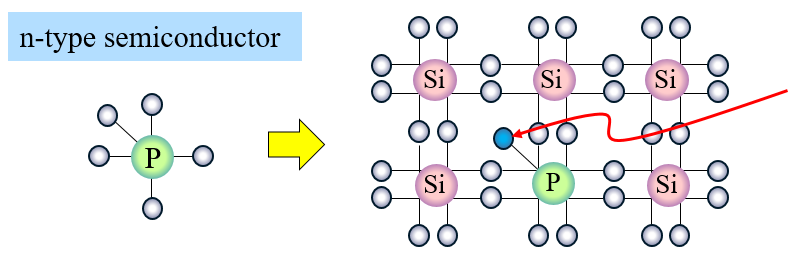
- Some of the silicon atoms are replaced with P (phosphorus).
-
This electron is “extra” in this structure.
This is a free electron.
When voltage is applied, it changes to a state where the electron is drawn to the positive (+) and can move freely (current flows).
-
-
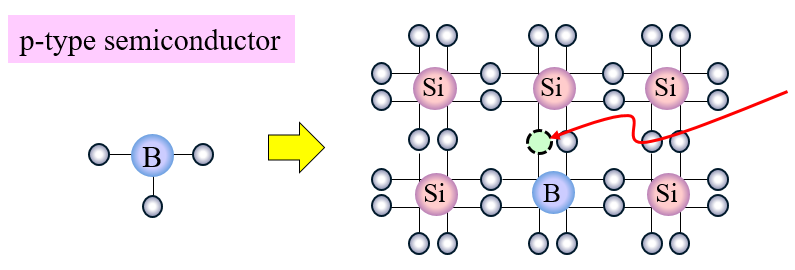
- Some of the silicon atoms are replaced with B (boron).
-
Here, this state leaves a “vacancy” where there is no electron.
This vacancy is called a hole. That is, the hole can also be called a virtual particle because it is void.
How current flows in p-type semiconductors
-
-
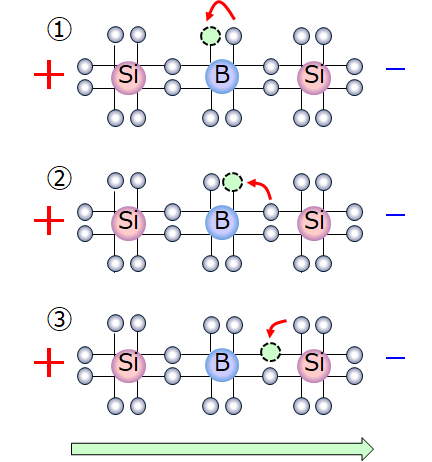
- Holes appear to be moving
-
Electrons are drawn to the positive (+) pole and move to close holes. Then, as the electrons move to vacant holes, new holes are created and then the adjacent electrons move to the new vacant holes again.
As this process is repeated, electrons move toward the positive (+) pole, and at the same time, the holes appear to move toward the negative (-) pole.
Only electrons are actually moving, but the holes can be considered as having positively charged particles.